PolyPid to Present New Phase 3 SHIELD II Data Demonstrating D-PLEX₁₀₀ Sustained Local Release at ESCMID Global 2026
PETACH TIKVA,
The abstract titled “Pharmacokinetics of local extended-release incisional doxycycline in patients undergoing abdominal colorectal surgery from the SHIELD II Phase 3 clinical trial,” was co-authored by Prof.
Key findings from the SHIELD II analysis:
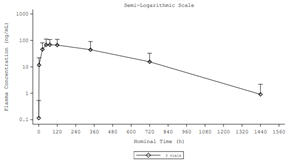
- Sustained and controlled release profile: Detectable doxycycline concentrations were observed for up to 763 hours (approximately 32 days), with no burst, following local application of D-PLEX₁₀₀ during surgery.
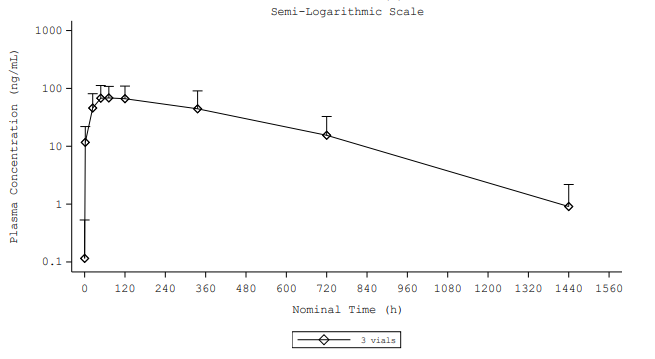
Figure 1. Mean (+ standard deviation) plasma doxycycline concentration versus time profile
- Minimal systemic exposure: Plasma doxycycline concentrations remained below 100 ng/mL, indicating negligible systemic levels relative to oral doxycycline administration where Cmax levels are approximately 16 to 26 times higher (1,613.3 ng/mL for 100 mg1 and 2,600 ng/mL for 200 mg2 doses).
“These pharmacokinetic results further support the mechanism of D-PLEX₁₀₀, which is designed to deliver sustained and controlled antibiotic exposure for 30 days directly at the surgical incision site,” said Dikla Czaczkes Akselbrad, Chief Executive Officer of
ESCMID Global 2026 Presentation Details
| Poster Title: | Pharmacokinetics of Local Extended-Release Incisional Doxycycline in Patients undergoing Abdominal Colorectal Surgery from the SHIELD II Phase 3 Clinical Trial |
| Presenter: | |
| Lead Author: | Prof. |
| Poster Details: | #P2740 (Presented at: Poster Hall B3) |
| Session: | 05b. Pharmacokinetics/pharmacodynamics of antibacterial drugs & therapeutic drug monitoring |
| Date/Time: |
The abstract will be available on www.polypid.com once the conference concludes.
About D-PLEX100
D-PLEX100, PolyPid’s lead product candidate, is designed to provide local prolonged and controlled anti-bacterial activity directly at the surgical site to prevent surgical site infections (“SSIs”). Following the administration of D-PLEX100 into the surgical site, PolyPid’s delivery technology, Kynatrix, pairs with Active Pharmaceutical Ingredients, enabling a prolonged and continuous release of the broad-spectrum antibiotic doxycycline, resulting in a high local concentration of the drug for a period of 30 days for the prevention of SSIs, with additional potential to prevent SSIs caused by antibiotic-resistant bacteria at the surgical site. D-PLEX100 recently demonstrated positive results in the Phase 3 SHIELD II trial, achieving a statistically significant 60% (p=0.0013) relative risk reduction in SSI incidence following abdominal colorectal surgery with large incisions. D-PLEX100 received Breakthrough Therapy designation from the U.S. Food and Drug Administration for the prevention of SSIs in patients undergoing elective colorectal surgery.
About
Forward-looking Statements
This press release contains “forward-looking statements” within the meaning of the Private Securities Litigation Reform Act and other securities laws. Words such as “expects,” “anticipates,” “intends,” “plans,” “believes,” “seeks,” “estimates” and similar expressions or variations of such words are intended to identify forward-looking statements. For example, the Company is using forward-looking statements when it discusses the potential mechanism and benefits of D-PLEX₁₀₀, including its ability to deliver sustained and controlled antibiotic exposure for 30 days directly at the surgical incision site, and the potential role of D-PLEX₁₀₀ in improving the prevention of surgical site infections. Forward-looking statements are not historical facts, and are based upon management’s current expectations, beliefs and projections, many of which, by their nature, are inherently uncertain. Such expectations, beliefs and projections are expressed in good faith. However, there can be no assurance that management’s expectations, beliefs and projections will be achieved, and actual results may differ materially from what is expressed in or indicated by the forward-looking statements. Forward-looking statements are subject to risks and uncertainties that could cause actual performance or results to differ materially from those expressed in the forward-looking statements. For a more detailed description of the risks and uncertainties affecting the Company, reference is made to the Company’s reports filed from time to time with the Securities and Exchange Commission, including, but not limited to, the risks detailed in the Company’s Annual Report on Form 20-F filed on February 25, 2026. Forward-looking statements speak only as of the date the statements are made. The Company assumes no obligation to update forward-looking statements to reflect actual results, subsequent events or circumstances, changes in assumptions or changes in other factors affecting forward-looking information except to the extent required by applicable securities laws. If the Company does update one or more forward-looking statements, no inference should be drawn that the Company will make additional updates with respect thereto or with respect to other forward-looking statements. References and links to websites have been provided as a convenience, and the information contained on such websites is not incorporated by reference into this press release. PolyPid is not responsible for the contents of third-party websites.
Company Contact:
908-858-5995
IR@Polypid.com
Investor Relations Contact:
Arx Investor Relations
North American Equities Desk
polypid@arxhq.com
1 Gschwend MH, Martin W, Erenmemişoğlu A, et. al.. Pharmacokinetics and Bioequivalence Study of Doxycycline Capsules in Healthy Male Subjects. Arzneimittelforschu 2007;57(6):347-51
2 Vibramycin (doxycycline hyclate) [capsules]: Full Prescribing Information.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/6016c990-606d-49e6-a7a8-4d88bace509a

Source: PolyPid Ltd.